Levo Gen 2 Treatment for Tinnitus
A therapy that reduces tinnitus and retrains the brain’s response
How does it work?
A comprehensive approach to managing tinnitus.
Our method—unique in the world and FDA-cleared in the United States—combines acoustic stimulation during sleep with a structured desensitization program to reduce the intensity, perception, and emotional impact of tinnitus.
How to get started
Who is a good candidate and how to begin therapy
The therapy can be delivered in-person, remotely, or through a hybrid approach, depending on your location. We always begin with a diagnostic evaluation to tailor the method to your auditory profile and set personalized expectations.
Schedule your initial evaluation
We’ll arrange your consultation in-person or remotely, based on your location and preference.
Diagnostic evaluation
A comprehensive clinical consultation, including audiological testing when indicated, based on local protocols.
Start of therapy
Access to Levo Gen 2, including your personalized acoustic prescription, usage guidance, and onboarding to our continuous learning platform.
Application and follow-up
Daily nighttime acoustic stimulation, combined with desensitization content delivered through our learning platform.
Includes at least 12 follow-up consultations to adjust your personalized acoustic prescription and monitor your clinical progress.
It includes at least 12 follow-up consultations with our clinical team, during which the individualized acoustic prescription is adjusted and your clinical progress is evaluated.
See if this therapy is right for you
Schedule your evaluation
Therapy stages
Acoustic prescription design
Creation of your personalized acoustic prescription tailored to your tinnitus profile
System learning and onboarding
Guided learning process to understand and effectively use the system
Continuous stimulation
Daily nighttime application of acoustic stimulation
Gradual withdrawal
Progressive reduction of stimulation to ensure long-lasting results
(Approximately 1 year)
The therapy can be delivered in-person, remotely, or through a hybrid approach, with professional support and continuous monitoring through our continuous learning platform, which is part of Levo.
Levo Gen 2
Precision biomedical technology designed for tinnitus management.
Each therapy begins with the design of a personalized acoustic prescription: a digitally generated sound that replicates the frequency, timbre, and intensity of the patient’s tinnitus.
This personalized signal is used during nighttime acoustic stimulation, promoting neuroplastic changes that progressively reduce the perception of the sound.
Continuous monitoring through the Levo Gen 2 platform
The platform records and visualizes progress, allowing the therapy to be adjusted to each patient’s needs:
-
Perceived intensity tracking (target outcome)
-
Subjective indicators: sleep quality, attention to tinnitus, and tolerance.
-
Adherence: hours of use and percentage of nights with acoustic stimulation.
Clinical note: Results and timelines may vary depending on the patient’s auditory profile, adherence, and comorbidities. The clinical team will guide the process individually and adjust the protocol based on each patient’s needs.
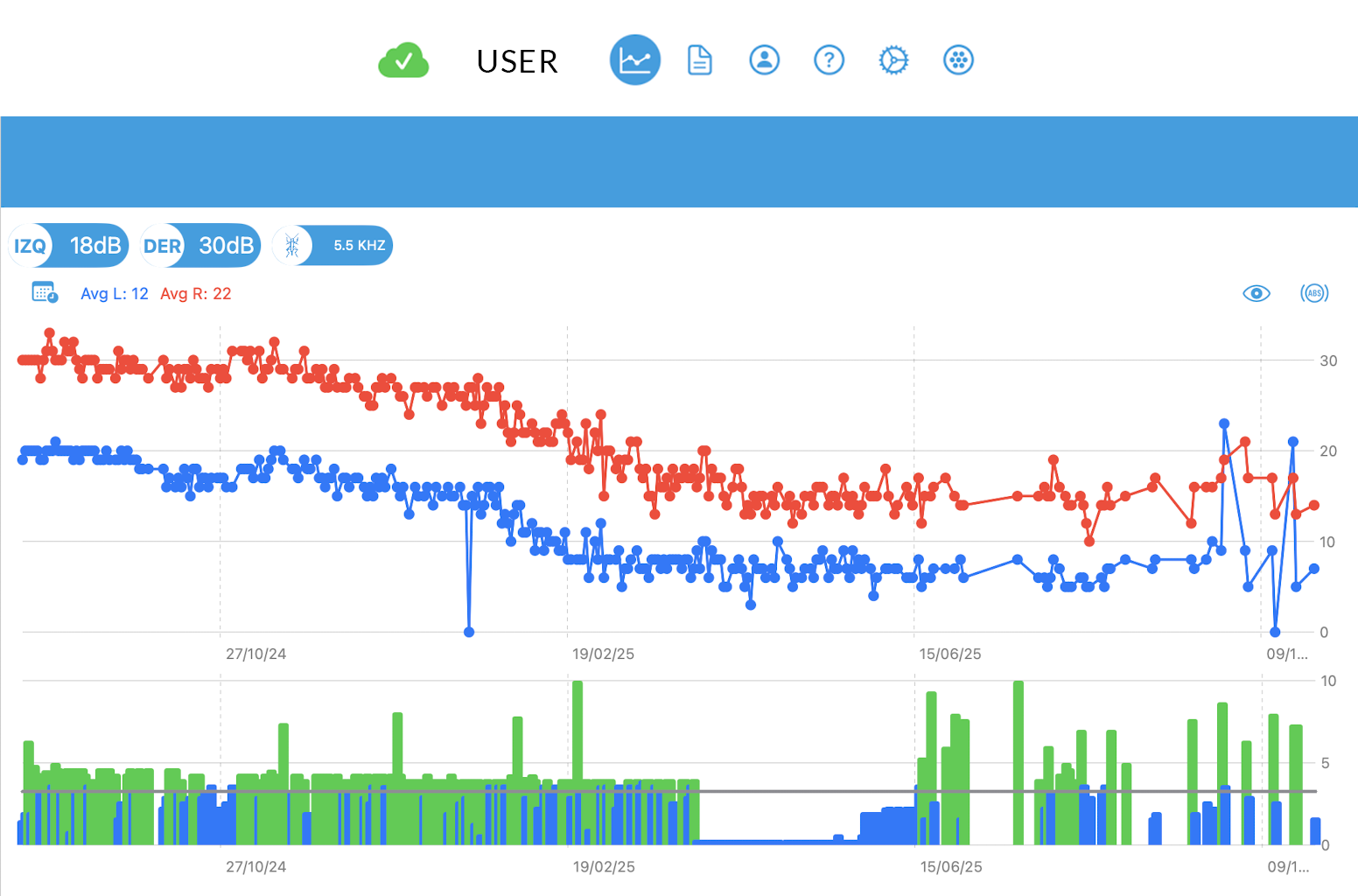
Perceived intensity
Reduction in tinnitus intensity in a patient over the course of one year of therapy.
Intensity progression chart over the therapy period. Each point represents the stimulation intensity used each night (blue: left ear / red: right ear).
The lower section shows the number of hours the patient used the device each night (green: >4 hours, blue: <4 hours).

Progress profile
Behavior of the different variables that make up the patient’s progress profile.
Each axis represents a dimension evaluated during the follow-up of the Levo Gen 2 therapy, and the shaded area reflects the level of progress in each one.
📊 Included variables
- Level: overall degree of progress within the therapy
- Consistency: regularity and adherence to system use
- Faces: subjective assessment of the patient’s perceived well-being or relief
- THI and TFI: internationally validated questionnaires that measure the impact on quality of life
- Sleep: a validated questionnaire that assesses sleep quality and the interference of tinnitus during sleep
- Stage: current phase of the therapeutic protocol
- Hours: average daily hours of device use
The progress score (23.72 in this example) integrates all these parameters into a single quantitative value, allowing clinicians to monitor the patient’s overall progress throughout the therapy.
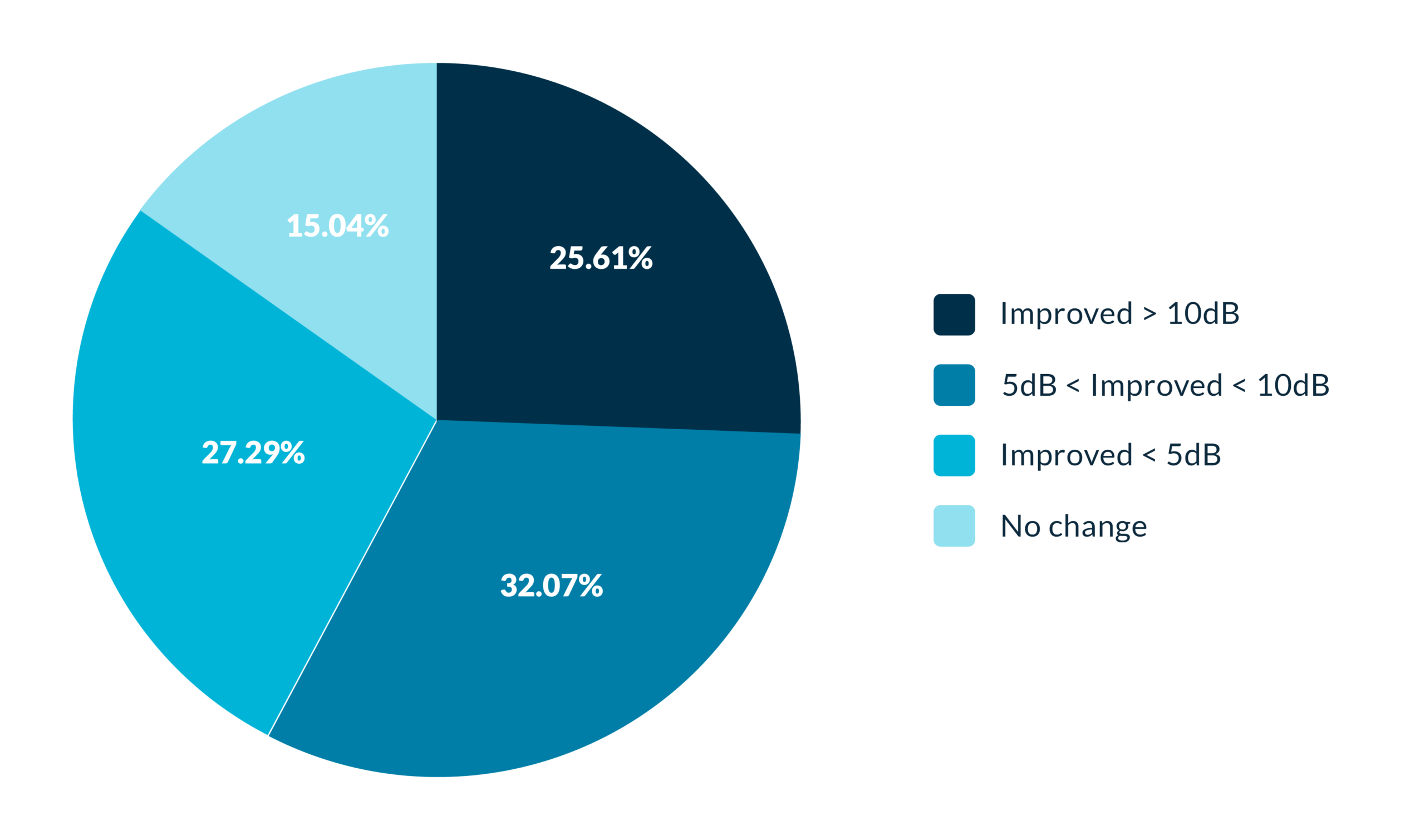
Overall outcomes
Therapy outcomes after 9 months (60 patients)
A retrospective study was conducted on 60 patients who completed the Levo Gen 2 therapy, evaluating the reduction in perceived tinnitus intensity at the end of the protocol (average follow-up of 9 months).
The chart shows the distribution of patients according to the degree of improvement in tinnitus intensity, measured in decibels (dB).
Clinical note:
This analysis focuses exclusively on changes in tinnitus intensity and does not assess improvements in quality of life.
The group of patients who did not show an objective reduction in intensity (15%) may still have experienced subjective clinical improvements—such as better sleep, habituation, or reduced distress—that are not reflected in this measurement.
This monitoring system supports both clinicians and patients by enabling continuous therapy tracking and ensuring a dynamic, personalized, and clinically validated therapeutic process.
FAQ
What can I expect from the therapy? Will my tinnitus go away?
The goal of the therapy is not to eliminate tinnitus completely, but to significantly reduce how it is perceived and how much it affects your daily life. Most patients experience a progressive decrease in intensity and distress, along with improved sleep and overall well-being. Results develop gradually over time.
How long does the therapy take?
The full therapy process typically lasts around 12 months.
This includes an initial phase of personalization and adaptation, followed by consistent use and gradual adjustment of the therapy based on your progress.
When will I start noticing results?
Results are gradual and vary from person to person.
Some patients begin to notice changes within the first few weeks, while for others it may take longer. Consistency is key to achieving meaningful and lasting improvements.
Do I need to listen to the therapy every day?
Yes. The therapy is designed to be used daily, primarily during sleep.
Regular use is essential to promote the neuroplastic changes that help reduce tinnitus perception.
Contact
Briefly tell us about your case, and a member of our clinical team will get back to you to coordinate the first step.
What happens after you submit the form?
-
We review your submission and propose available time slots for your evaluation.
-
If appropriate, we send you an initial questionnaire to better understand your hearing profile.
-
We guide you on the consultation format (in-person or remote) and the next steps.